Science and big pharma are joined at the hip. Does that skew results?
Last November, Novo Nordisk released a study that found its weight-loss drug semaglutide reduced the risk of a heart attack, stroke or cardiovascular death.
So what? All 16 researchers on the study declared interests in the company.
Pharma companies funding clinical trials – and the researchers who work on them – are an inevitable part of drug research and development. That said…
- By Novo Nordisk’s own admission, profits from the booming growth in the market for GLP-1 agonists (like semaglutide) are unprecedented.
- The company is currently suspended from the UK’s pharma trade body for not being transparent in its promotion of semaglutide’s weight-loss predecessor, liraglutide, to doctors.
- Researchers say conflicts of interest can put patient safety and appropriate use of government healthcare funding at risk.
The products. Novo Nordisk makes two injectable products with the active ingredient semaglutide: Ozempic and Wegovy. The former is designed for diabetics and used off-label for weight loss. The latter is higher dosage and is approved specifically for weight loss.
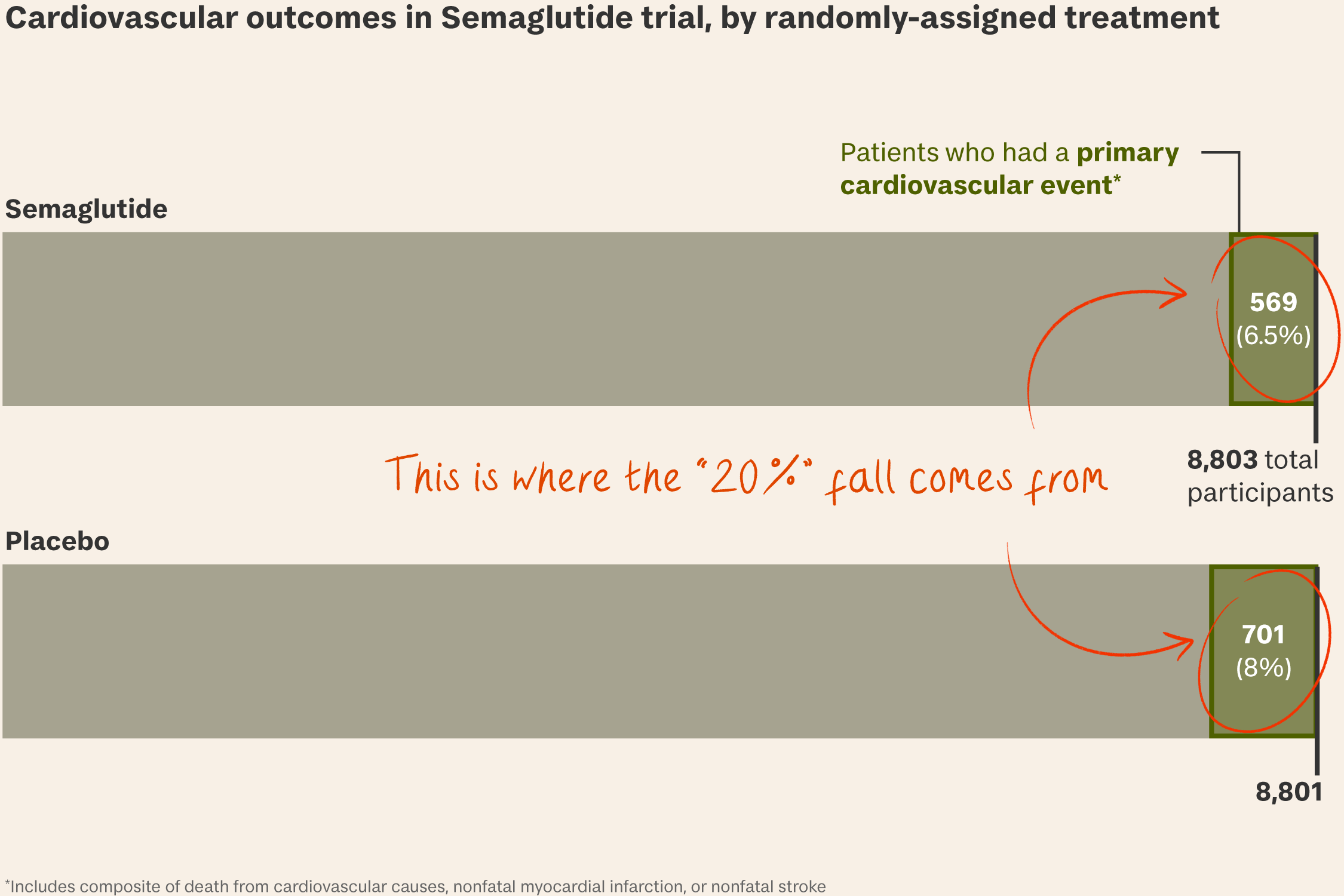
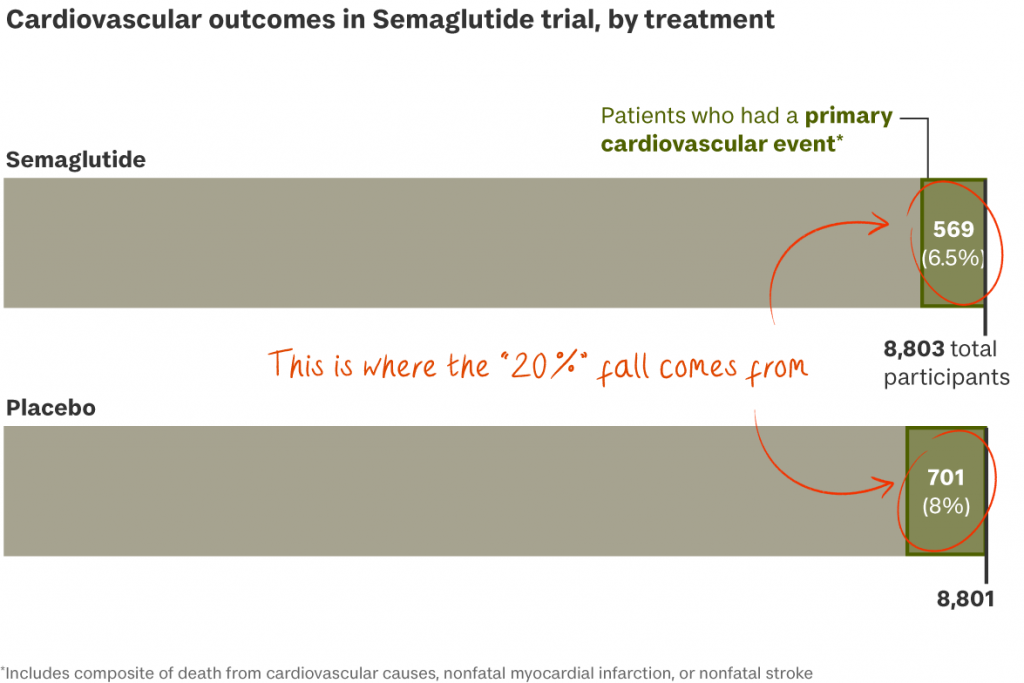
The SELECT trial. Last August, the Danish company issued a press release claiming a 2.4 mg dose of semaglutide (Wegovy) could reduce the risk of “major adverse cardiovascular events” by 20 per cent in overweight or obese adults, boosting its stock price by 17 per cent.
- Last November the full study behind the claim showed that 6.5 per cent of patients who took semaglutide “experienced death from cardiovascular causes, nonfatal myocardial infarction, or nonfatal stroke”, compared with 8 per cent of patients taking a placebo.
- In May the study’s lead author, Professor John Deanfield, presented further unpublished findings at an obesity conference in Venice (run by a group part-funded by Novo Nordisk) claiming semaglutide led to cardiovascular benefits irrespective of weight loss.
Deanfield’s findings led to media coverage on the potential wider benefits of semaglutide, including a front-page story in The Times in which he claimed: “a large number of potential patients who have cardiovascular disease would benefit from use of this drug”.
The disclosures. Tortoise reviewed the researchers’ disclosures attached to the November 2023 study and found:
- Seven of the authors work for Novo Nordisk and have stock options or stock in the company, although their salaries and value of the stock have not been made public.
- The other 11 researchers all declared some form of consultancy, advisory, travel, research or speaker role for Novo Nordisk.
- Overall, according to public disclosure databases in the UK and US, those 11 researchers received more than $7.5 million from Novo Nordisk between 2015 and 2022.
Deanfield reported over £34,500 in payments from Novo Nordisk between 2020 and 2022 to the Association of the British Pharmaceutical Industry (ABPI).
Dr Donna Ryan, a former president of the Obesity Society in the US, has declared more than $700,000 in payments since 2015. Dr Ildiko Lingvay, a member of the American Diabetes Association, has declared over $5.3 million in associated research funding from Novo.
Not the whole picture. Dr Piotr Ozieranski, who researches conflicts of interest at the University of Bath, analysed the ABPI’s disclosure database and reports published on Novo’s Nordisk’s website, which found:
- Between 2015 and 2021 the company more than doubled its spending on research and development in the UK from £5.3 million to over £13 million. This includes payments to unnamed healthcare professionals and organisations.
- Novo also increased payments to named healthcare organisations, professionals and patient organisations from £5.7 million to £8.1 million.
This doesn’t mean the studies based on the SELECT trial are unreliable or the results are inaccurate. The researchers all declared their interests and there is nothing to suggest they were unduly influenced.
But… Ozieranski says there are “clear and proven risks” associated with conflicts of interest which “can have a detrimental impact on patient health and safety” if medical professionals prioritise their interests over providing care.
What’s more… Novo Nordisk is on track to make Ozempic and Wegovy some of the most prescribed drugs in history before its patent for semaglutide runs out in the early 2030s. It’s also funding research into the potential benefits of semaglutide for treating kidney disease and dementia.
Tortoise approached all the independent study researchers for comment but did not receive responses from those named.
In a statement, Novo Nordisk said researchers clearly disclose their relationships with the company and that it works with external researchers and experts “to ensure the studies are conducted with the highest standards and ultimately meet patient needs”.